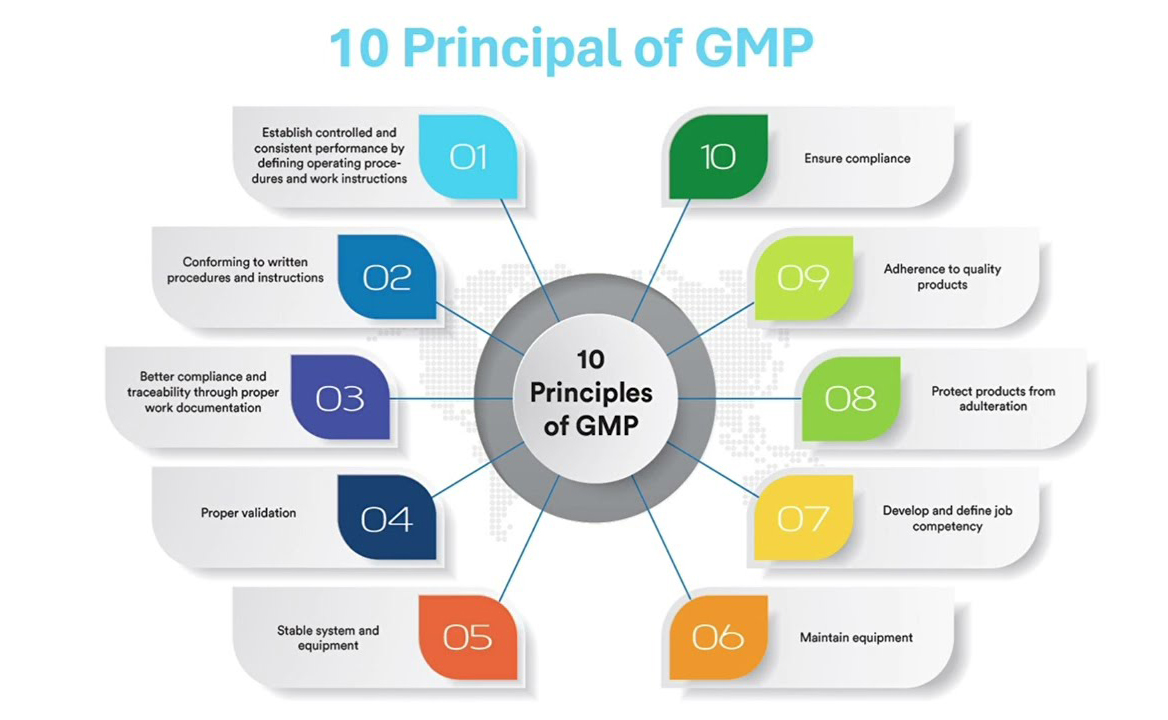
Symyza Biotech . has announced significant enhancements to its quality assurance and compliance systems, reaffirming its commitment to maintaining the highest standards in pharmaceutical manufacturing.
The upgraded systems include advanced quality control processes, improved documentation practices, and enhanced monitoring mechanisms. These measures are designed to ensure full compliance with regulatory requirements and industry best practices.
The company has invested in modern laboratory equipment and digital quality management systems to support accurate testing and efficient data management. These improvements will enable Symyza Biotech to maintain consistency in product quality and ensure patient safety.
A company representative noted that quality is at the core of Symyza’s operations. “Our focus on quality and compliance is unwavering. We continuously strive to improve our processes and systems to meet evolving industry standards,” the representative said.
By strengthening its quality infrastructure, Symyza Biotech aims to build trust with its customers and stakeholders while reinforcing its reputation as a reliable pharmaceutical manufacturer.